Sepifilm LP provides excellent moisture protection which is crucial when working with moisture sensitive actives in oral solid dosage forms
In the field of formulation, one of the basic requirements is that the active ingredient remains stable in the finished dosage form until the end of its shelf life to ensure efficacy and patient safety. The degradation of the active can occur through thermal degradation, oxidation, UV, microorganisms or hydrolysis. Atmospheric humidity is one of the main sources of moisture that influences the active ingredient chemically or physically.
Protecting the cores with a moisture barrier film is an appropriate solution. An ideal moisture barrier coating should exhibit low permeability to water vapour without compromising its dissolution functionality. For a film-coating to be able to protect the core, it must contain a hydrophobic ingredient. This ingredient can be the polymer itself or a hydrophobic plasticiser.
SEPIFILM™ LP product characteristics
SEPPIC’s Sepifilm™ LP range has been used for over 30 years for the purposes of moisture protection in pharmaceutical as well as nutrition applications. Sepifilm™ LP is a ready-to-use film coating range of excipients containing stearic acid that prevents the moisture from reaching the core of the dosage form.
Sepifilm™ LP products can be applied on powder, pellets or tablets at varying weight gains depending on the protection level to be achieved.
SEPIFILM™ LP performances
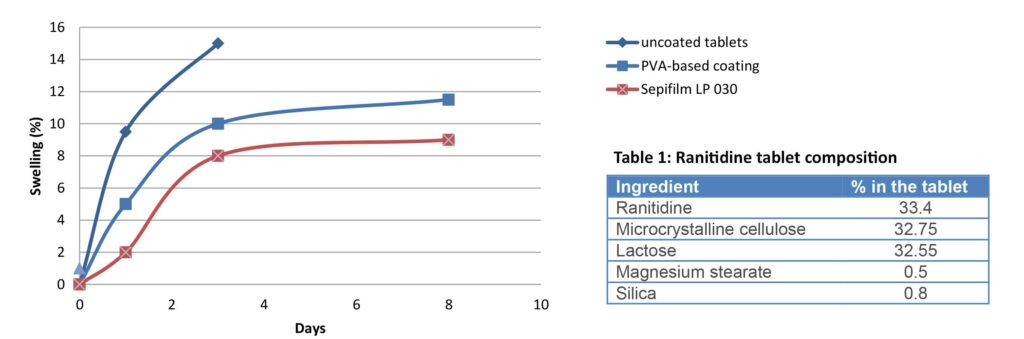
500 mg Ranitidine tablets (Table 1) were coated with various ready-to-use solutions in order to test the products’ performance regarding moisture protection. PVA-based coating designed for moisture protection was compared to Sepifilm™ LP 030. 5% weight gains were applied for all film-coating using the vendors’ recommendation for application, and the swelling of the tablets was measured during 8 days at 25°C and 75% RH.
The results in Graph 1 show that Ranitidine tablets coated with Sepifilm™ LP 030 swelled less than the other tablets, indicating that the coating prevents moisture from entering the core. Uncoated tablets became too soft to be handled after 3 days.
Sepifilm™ LP performance was also demonstrated using Dynamic Vapor Sorption (DVS), a gravimetric technique that measures how quickly and how much of a solvent is absorbed by a sample such as dry powder absorbing water.
Tablets containing SEPIFIT™ Protect, a hygroscopic nutraceutical active (Table 2) containing polyphenols, were coated with various ready-to-use solutions in order to test the products’ performance regarding moisture protection.
Tablets were placed in the DVS measuring device. The percentage of relative humidity was gradually increased from 0%RH up to 90%RH (sorption cycle) and then decreased (desorption cycle). The weight variation of the tablet was recorded continuously thanks to a microbalance.
The maximum weight variation is an important parameter because it reflects the water uptake of the tablet. The final weight variation is the second parameter taken into account since it represents the amount of water trapped inside the core involving the risk of hydrolysing the active.

Uncoated tablets adsorb much more moisture than coated ones but they dry more as well. On the contrary, coated tablets absorb less moisture but some water remains inside the core. The reason for this is that the coating forms a barrier preventing water from entering the core. However, the coating process involves the use of water, and a certain amount of water is bound to enter the core. The aim of moisture protection film-coating is to include as little water as possible.
The DVS recording showed that tablets coated with Sepifilm™ LP 030 absorb less water than PVA-based coated tablets, and moreover the remaining water content is lower, making Sepifilm™ LP 030 the best choice for moisture protection.
It is possible to combine the moisture protection coating with an anticaking agent such as Sepistab™ ST 200 to improve the formulation.
SEPPIC and Alsiano are your partners of choice for the formulation of moisture sensitive actives in Oral Solid Dosage Forms. Thanks to many years of expertise we can provide data, trainings, technical visits as well as all documents required for the registration of the finished dosage form.


